|
In other words, each element has a unique number that identifies how many protons are in one atom of that element. The number of protons in the nucleus of an atom determines an element's atomic number. Define and determine the mass number of an atom.Define and determine the atomic number of an atom.Hydrogen – Properties Summary Element Hydrogen Atomic Number 1 Symbol H Element Category Non Metal Phase at STP Gas Atomic Mass 1.0079 Density at STP 0.0899 Electron Configuration 1s1 Possible Oxidation States +1,-1 Electron Affinity 72.8 Electronegativity 2.2 1st Ionization Energy 13.5984 Year of Discovery 1766 Discoverer Cavendish, Henry Thermal properties Melting Point -259.1 Boiling Point -252.9 Thermal Conductivity 0.1805 Specific Heat 14.304 Heat of Fusion 0.05868 Heat of Vaporization 0.After reading this section you will be able to do the following: The Standard English unit is pounds mass per cubic foot ( lbm/ft 3). The standard SI unit is kilograms per cubic meter ( kg/m 3). In other words, the density (ρ) of a substance is the total mass (m) of that substance divided by the total volume (V) occupied by that substance. It is an intensive property, which is mathematically defined as mass divided by volume: Typical densities of various substances at atmospheric pressure.ĭensity is defined as the mass per unit volume. How does the atomic mass determine the density of materials? Density of Hydrogen 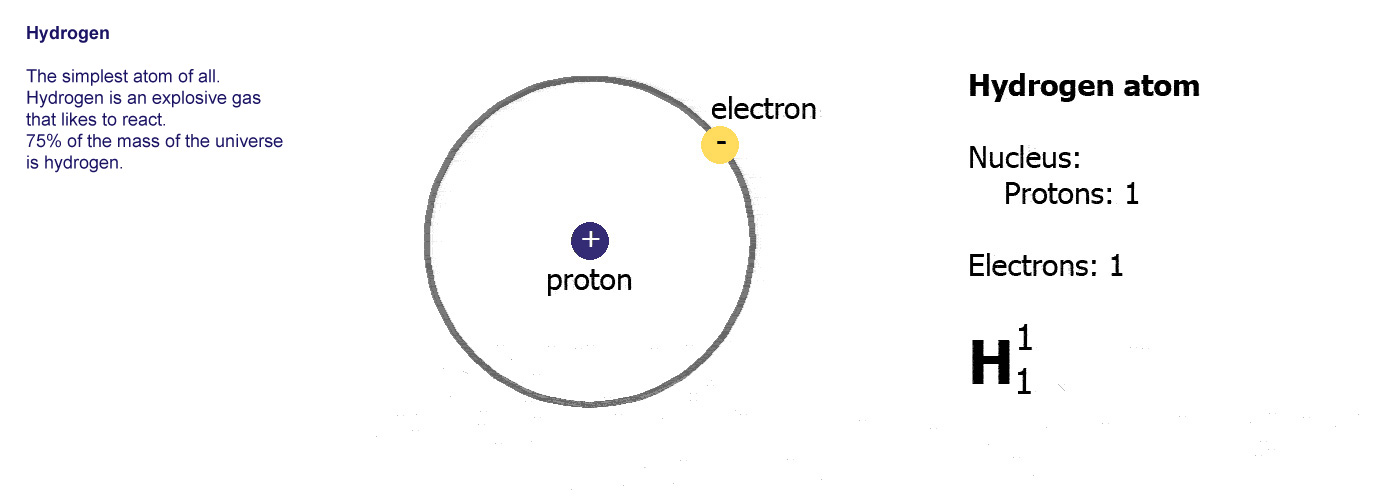
The atomic mass number determines especially the atomic mass of atoms. The mass number is different for each different isotope of a chemical element. For 63Cu, the atomic mass is less than 63, so this must be the dominant factor. A nucleus with greater binding energy has lower total energy, and therefore a lower mass according to Einstein’s mass-energy equivalence relation E = mc 2. The nuclear binding energy varies between nuclei.  
This increases the mass of nuclei with more neutrons than protons relative to the atomic mass unit scale based on 12C with equal numbers of protons and neutrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
